To identify functional groups in IR spectrum, analyze the peaks at specific wavenumbers corresponding to different functional groups, such as carbonyl, hydroxyl, or amine groups. Additionally, compare the observed peaks with reference IR spectra or databases for accurate identification.
Understanding how to identify functional groups in IR spectra is essential for organic chemists and researchers. Infrared (IR) spectroscopy is a valuable tool for analyzing the chemical structure of organic compounds by measuring the vibrational frequencies of their functional groups.
By interpreting the peaks in an IR spectrum, it is possible to determine the presence of specific functional groups, such as carbonyl, hydroxyl, or amine groups. This information is crucial for identifying unknown compounds or verifying the structure of known compounds. We will explore the key steps and techniques for identifying functional groups in IR spectra, enabling readers to enhance their skills in organic compound analysis.
Essentials Of Ir Spectrum Analysis
Understanding the essentials of IR spectrum analysis is crucial for identifying functional groups in the infrared (IR) spectrum. The analysis involves recognizing the characteristic absorption bands of different functional groups, providing valuable insights into the chemical structure of the molecule under investigation. Here, we will delve into the fundamental aspects of IR spectrum analysis, emphasizing the importance of sample preparation and highlighting common pitfalls to avoid when interpreting IR spectra.
Understanding The Basics Of An Ir Spectrum
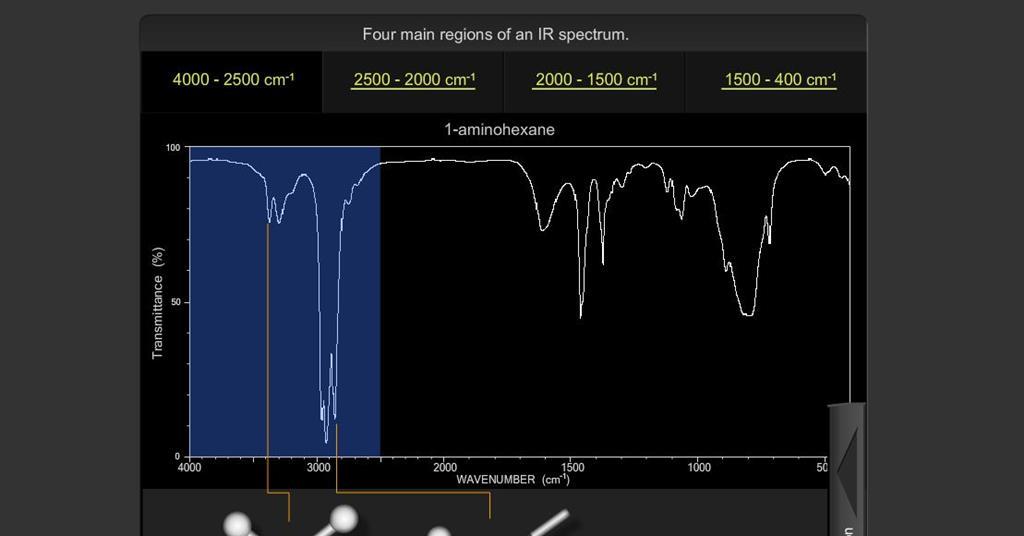
When analyzing an IR spectrum, it is essential to comprehend the fundamental principles underlying the technique. IR spectroscopy measures the interactions between infrared radiation and the molecular vibrations of a sample, resulting in distinct absorption bands that correspond to specific functional groups. These absorption bands are displayed as peaks on the IR spectrum, each indicative of the presence of a particular functional group. By understanding the characteristic wavenumber ranges associated with different functional groups, analysts can confidently identify and interpret the peaks in an IR spectrum.
The Importance Of Sample Preparation
Sample preparation plays a pivotal role in IR spectrum analysis, as the quality of the sample directly impacts the accuracy and reliability of the results. Proper preparation involves creating a homogeneous thin film of the sample to ensure consistent and reliable spectra. Additionally, attention to detail in sample handling and mounting is crucial to minimize artifacts and interference, thereby enhancing the clarity of the spectral data. By adhering to rigorous sample preparation protocols, analysts can minimize the potential for errors and obtain precise infrared spectra for accurate functional group identification.
Common Pitfalls To Avoid In Ir Spectrum Analysis
While conducting IR spectrum analysis, it is imperative to be aware of common pitfalls that may impede accurate interpretation. Some of these pitfalls include incomplete sample drying, improper instrument calibration, and overlooking spectral artifacts such as water vapor and carbon dioxide absorptions. Additionally, overlooking the presence of impurities or contaminants in the sample can lead to misinterpretation of spectral features. By diligently addressing these potential pitfalls and incorporating robust quality control measures, analysts can ensure the reliability and integrity of their IR spectrum analysis results.
Credit: edu.rsc.org
How To Identify Key Functional Groups
When analyzing an IR (Infrared) spectrum, it’s essential to be able to identify key functional groups. These key functional groups are indicative of specific chemical bonds and structures, which can be crucial in determining the composition of a compound. Here’s a guide on how to identify these key functional groups in an IR spectrum.
Recognizing Signature Peaks And Their Frequencies
One of the most effective methods of identifying key functional groups in an IR spectrum is to recognize the signature peaks and their associated frequencies. Each functional group exhibits characteristic absorption peaks at specific frequencies, which can provide valuable insights into the compound’s structure.
- Carbonyl Group (C=O): The carbonyl group typically appears as a strong, sharp peak in the range of 1680-1750 cm-1.
- Hydroxyl Group (OH): Hydroxyl groups are identifiable by a broad peak in the region of 3200-3600 cm-1.
- Amine Group (NH2): Amines often display peaks in the range of 3300-3500 cm-1 for primary amines and 3100-3500 cm-1 for secondary amines.
Utilizing Peak Tables And Databases
Another valuable approach to identifying functional groups in an IR spectrum is to utilize peak tables and databases. Various reference materials provide comprehensive lists of characteristic peaks associated with specific functional groups, aiding in the interpretation of spectral data.
| Functional Group | Characteristic Peak Frequency (cm-1) |
|---|---|
| Alkane C-H Stretch | 2850-2960 |
| Aromatic C=C Stretch | 1600-1580 |
| Carbonyl C=O Stretch | 1680-1750 |
By referencing these tables and databases, analysts can quickly identify the presence of specific functional groups based on the observed peak frequencies in the IR spectrum.
Interpreting Ir Spectrum Patterns
Interpreting IR spectrum patterns is crucial in identifying functional groups present in a compound. This process involves analyzing peak shapes, understanding distinctions between overtone and combination bands, and recognizing prominent signals indicating specific functional groups.
Analyzing Peak Shapes And Their Variations
When analyzing peak shapes in the IR spectrum, it’s essential to note the characteristics associated with different functional groups. Sharp and intense peaks typically indicate the presence of strongly bonded functional groups, such as the C=O bond in ketones and aldehydes. Conversely, broad and not very intense peaks are often associated with weakly bonded functional groups, like those containing hydrogen bonding or conjugation.
Additionally, variations in peak shapes, such as asymmetry or shoulders, can provide insight into the environmental factors affecting the functional groups, including steric effects or hydrogen bonding. Understanding these nuances can help in narrowing down potential functional groups within the compound.
Distinction Between Overtone And Combination Bands
When examining an IR spectrum, it’s essential to distinguish between overtone and combination bands. Overtone bands occur at approximately two times the frequency of the fundamental vibrations and are typically much weaker in intensity. In contrast, combination bands result from the simultaneous excitation of multiple vibrational modes within a molecule, leading to complex peak patterns in the spectrum.
Recognizing the distinction between overtone and combination bands is crucial for accurately interpreting the IR spectrum and identifying the presence of certain functional groups. Additionally, understanding the expected frequencies for these bands can aid in differentiating between them and ruling out potential misinterpretations.
Tips For Complex Sample Analysis
When analyzing complex samples using IR spectroscopy, identifying functional groups can be challenging. However, by applying the right techniques, it is possible to effectively analyze complex samples and identify functional groups accurately. Here are some valuable tips for complex sample analysis.
Dealing With Overlapping Peaks
One common challenge in identifying functional groups in complex samples is dealing with overlapping peaks in the IR spectrum. When multiple functional groups are present, their absorption peaks may overlap, making it difficult to distinguish between them. To address this issue, it is essential to carefully analyze the entire spectrum, paying close attention to the shape and intensity of the peaks. Additionally, utilizing advanced spectral deconvolution techniques can help separate overlapping peaks, enabling the accurate identification of individual functional groups.
Identifying Functional Groups In Multimolecular Systems
Complex samples often consist of multimolecular systems, where multiple compounds contribute to the overall IR spectrum. When analyzing such samples, it is crucial to employ spectral subtraction methods to isolate the spectrum of the target molecule or functional group. By subtracting the spectrum of other components present in the sample, the unique absorption features of the functional groups can be identified with greater precision.
Utilizing Software In Ir Analysis
Software Tools That Enhance Functional Group Identification
Software tools play a crucial role in enhancing the accuracy and efficiency of functional group identification in IR spectroscopy analysis. By utilizing advanced software, researchers and analysts can streamline the process to identify and interpret functional groups present in the sample.
How To Use Software For Better Accuracy In Analysis
Software tools such as ChemDraw, IR-spectra interpretation software, and SpectraGryph are widely utilized in the analysis of IR spectra for functional group identification. These tools facilitate the comparison of experimental and reference spectra, allowing for better accuracy in pinpointing the characteristic peaks associated with specific functional groups.
Moreover, the implementation of software enables the generation of precise peak picking and baseline correction, leading to improved peak assignments and overall analysis accuracy.
Enhancing Your Ir Spectrum Interpretation Skills
Enhancing your IR spectrum interpretation skills involves mastering the identification of functional groups. By understanding the characteristic peaks and patterns in IR spectra, you can skillfully recognize functional groups, enabling accurate compound identification and analysis. This proficiency is vital for various scientific and industrial applications.
Advanced Techniques For Identifying Subtle Differences
Enhancing your IR spectrum interpretation skills involves delving into advanced techniques that allow for the identification of subtle differences within functional groups. By understanding these intricacies, you can elevate your ability to pinpoint specific functional groups and gain a more comprehensive understanding of the molecular structure being analyzed.
- Utilize computer software that can compare and analyze spectra to identify subtle differences.
- Experiment with different sample preparation techniques to enhance resolution and reveal hidden spectral features.
- Explore advanced spectral manipulation methods such as derivative spectroscopy to highlight subtle spectral changes.
Continuous Learning And Staying Updated With New Research
Continuous learning plays a crucial role in refining your IR spectrum interpretation skills. Staying updated with the latest research and developments in the field of infrared spectroscopy enables you to adapt to new methodologies and expand your knowledge base.
- Engage with professional networks and attend conferences to stay informed about cutting-edge research in the field.
- Regularly review scientific journals and publications to keep abreast of advancements in IR spectroscopy techniques and methodologies.
- Participate in workshops and webinars conducted by industry experts to gain insights into novel approaches for interpreting IR spectra.
Case Studies: Applied Functional Group Identification
In case studies focusing on applied functional group identification, the key topic revolves around the methodology for identifying functional groups in IR spectra. By employing specific techniques and analysis, researchers can effectively pinpoint and interpret functional groups present in the IR spectrum, allowing for a more comprehensive understanding of molecular structures and chemical compositions.
Real-world Examples Of How To Identify Functional Groups In Ir Spectrum
Functional group identification in IR spectrum analysis can be challenging, and real-world examples can provide valuable insights into the process. Let’s take a look at some case studies that demonstrate the application of functional group identification in IR spectroscopy.
Lessons Learned From Common Mistakes In Analysis
When analyzing IR spectra for functional groups, it’s crucial to learn from common mistakes that can occur. By understanding these pitfalls, analysts can improve their accuracy and ensure reliable results in functional group identification.
Essential Tips For Ir Analysis Success
Best Practices Recap For Identifying Functional Groups Effectively
When analyzing IR spectra for functional groups, it’s essential to follow best practices to ensure accurate identification. Here’s a recap of the most crucial tips:
- Understand the basics: Make sure you have a solid understanding of the fundamental principles of IR spectroscopy and functional groups.
- Use reference materials: Utilize reliable reference materials such as IR spectral databases, textbooks, and online resources to aid in the interpretation of IR spectra.
- Focus on key regions: Pay close attention to specific regions of the IR spectrum, such as the fingerprint region and characteristic absorption bands for different functional groups.
- Practice pattern recognition: Train your eye to recognize common patterns and peaks associated with different functional groups, allowing for quicker and more accurate analysis.
Ongoing Practices To Refine Analysis Expertise
Refining your expertise in IR analysis requires continuous effort and practice. Consider the following ongoing practices to enhance your skills:
- Engage in hands-on experience: Actively seek out opportunities to analyze real-world samples using IR spectroscopy to gain practical experience and improve your analytical capabilities.
- Seek feedback and collaboration: Collaborate with peers, mentors, or experts in the field to discuss and verify your interpretations of IR spectra, receiving valuable feedback to refine your analysis.
- Stay updated with advancements: Keep abreast of the latest developments in IR spectroscopy and functional group analysis through scientific journals, conferences, and workshops.
- Continuously validate interpretations: Regularly validate your interpretations of IR spectra by cross-referencing with known compounds and consulting with experienced professionals to enhance the accuracy of your analyses.
Frequently Asked Questions For How To Identify Functional Groups In Ir Spectrum
What Are Functional Groups In Ir Spectrum?
Functional groups in IR spectrum are specific patterns of peaks that indicate the presence of certain chemical groups. They provide valuable information about the molecular structure and can help identify compounds based on their unique spectral fingerprints.
How To Interpret Functional Groups From Ir Spectrum?
Interpreting functional groups from IR spectrum involves analyzing the peaks and their intensities to identify characteristic absorption bands associated with specific chemical groups. This process requires understanding the correlation between functional groups and their corresponding spectral features.
Why Is It Important To Identify Functional Groups In Ir Spectrum?
Identifying functional groups in IR spectrum is crucial for determining the structure of organic compounds. It helps in identifying unknown substances, verifying chemical composition, and understanding the reactivity and properties of molecules, which is essential in various scientific fields such as chemistry, pharmacology, and biochemistry.
Conclusion
Identifying functional groups in IR spectra is crucial for understanding molecular structure. By utilizing peak positions and intensities, chemists can determine specific groups present in a compound. Learning to interpret IR spectra can greatly aid in characterizing unknown compounds and advancing research in various fields.
Mastering this skill is indispensable for any chemistry enthusiast.

